Reaction Rates And Chemical Equilibrium Report Sheet - Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: To summarise, the rate of reaction can be affected by: A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. (1) temperature, (2) particle size, and (3) concentration of the reactants. When the rate of the forward and reverse of a reversible process are equal. Shifts to right (towards products), shifts to. Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be.
Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. (1) temperature, (2) particle size, and (3) concentration of the reactants. A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: When the rate of the forward and reverse of a reversible process are equal. Shifts to right (towards products), shifts to. To summarise, the rate of reaction can be affected by:
To summarise, the rate of reaction can be affected by: (1) temperature, (2) particle size, and (3) concentration of the reactants. When the rate of the forward and reverse of a reversible process are equal. Shifts to right (towards products), shifts to. Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,.
Solved REPORT SHEET Reaction Rates and Chemical Equilibrium
A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. Shifts to right (towards products), shifts to. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: When the rate of the forward and reverse of a reversible process are equal. (1) temperature, (2).
Chemical Equilibrium Report Sheet
(1) temperature, (2) particle size, and (3) concentration of the reactants. Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. Shifts to right (towards products), shifts to. When the rate of the forward and reverse of a reversible process are equal. To summarise, the rate of.
Solved Date Team Instructor REPORT SHEET Reaction Rates and
A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. Shifts to right (towards products), shifts to. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: To summarise, the rate of reaction can be affected by: Exercise we will explore the concept of.
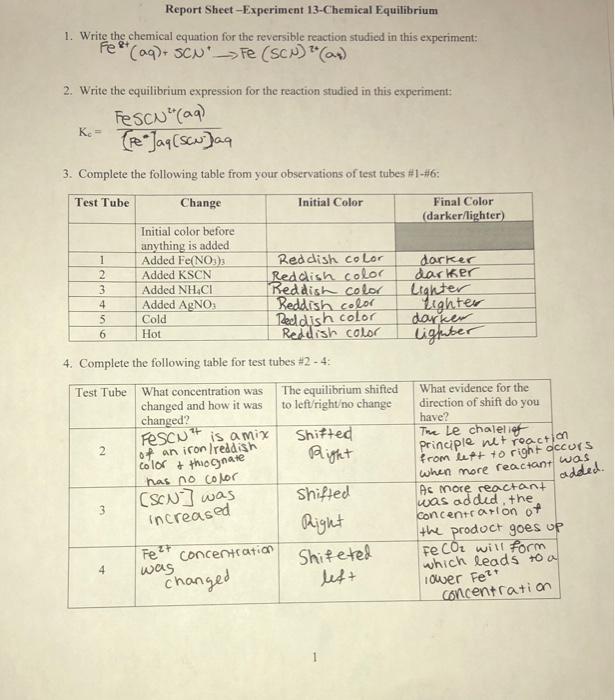
Solved Report SheetExperiment 13Chemical Equilibrium 1.
Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: Shifts to right (towards products), shifts to. To summarise, the rate of reaction can be affected by: Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. A catalyst increases the.
Solved Reaction Rates and Equilibrium Report Sheet Date
Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: (1) temperature, (2) particle size, and (3) concentration of the reactants. A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. When the rate of the forward and reverse of a reversible process are.
CHEMISTRY O LEVEL(FORM THREE) NOTES CHEMICAL EQUILIBRIUM
Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: (1) temperature, (2) particle size, and (3) concentration of the reactants. To summarise, the rate of reaction can be affected by: Shifts to right (towards products), shifts to. When the rate of the forward and reverse of a reversible process are equal.
Solved REPORT SHEET Reaction Rates and Chemical Equilibrium
When the rate of the forward and reverse of a reversible process are equal. A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. Shifts to right (towards products), shifts to. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: (1) temperature, (2).
Chemical Equilibrium Worksheet 1
Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. A catalyst increases the rate of a chemical reaction by lowering the activation energy required for the reaction to occur,. (1) temperature, (2) particle size, and (3) concentration of the reactants. To summarise, the rate of reaction.
SOLVED REPORT SHEET Reaction Rates and Chemical Equilibrium LAB 18 A
Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. To summarise, the rate of reaction can be affected by: (1) temperature, (2) particle size, and (3) concentration of the reactants..
Solved Reaction Rates & Chemical Equilibrium Name Report
To summarise, the rate of reaction can be affected by: Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: When the rate of the forward and reverse of a reversible process are equal. (1) temperature, (2) particle size, and (3) concentration of the reactants. Shifts to right (towards products), shifts to.
(1) Temperature, (2) Particle Size, And (3) Concentration Of The Reactants.
When the rate of the forward and reverse of a reversible process are equal. Exercise we will explore the concept of equilibrium, or how far a reaction proceeds toward the formation product, and what factors can be. Using le chatelier’s principle, predict how each of the changes would affect the equilibrium systems ex: To summarise, the rate of reaction can be affected by:
A Catalyst Increases The Rate Of A Chemical Reaction By Lowering The Activation Energy Required For The Reaction To Occur,.
Shifts to right (towards products), shifts to.